3 Interoceptive inference, emotion, and predictive selfhood
3.1 Interoceptive inference and emotion
Considering the cybernetic roots of PP, together with the free energy principle, leads to a potentially counterintuitive idea. This is that PP may apply more naturally to interoception (the sense of the internal physiological condition of the body) than to exteroception (the classic senses, which carry signals that originate in the external environment). This is because for an organism it is more important to avoid encountering unexpected interoceptive states than to avoid encountering unexpected exteroceptive states. A level of blood oxygenation or blood sugar that is unexpected is likely to be bad news for an organism, whereas unexpected exteroceptive sensations (like novel visual inputs) are less likely to be harmful and may in some cases be desirable, as organisms navigate a delicate balance between exploration and exploitation (Seth 2014a), testing current perceptual hypotheses through active inference (see section 5, below), all ultimately in the service of maintaining organismic homeostasis.
Perhaps because of its roots in Helmholtz, PP has largely been developed in the setting of visual neuroscience (Rao & Ballard 1999), with a related but somewhat independent line in motor control (Wolpert & Ghahramani 2000). Recently, an explicit application of PP to interoception has been developed (Seth 2013; Seth & Critchley 2013; Seth et al. 2011; see also Gu et al. 2013). On this theory of interoceptive inference (or equivalently interoceptive predictive coding), emotional states (i.e., subjective feeling states) arise from top-down predictive inference of the causes of interoceptive sensory signals (see Figure 4). In direct analogy to exteroceptive PP, emotional content is constitutively specified by the content of top-down interoceptive predictions at a given time, marking a distinction with the well-studied impact of expectations on subsequent emotional states (see e.g., Ploghaus et al. 1999; Ueda et al. 2003). Furthermore, interoceptive prediction errors can be minimized by (i) updating predictive models (perception, corresponding to new emotional contents); (ii) changing interoceptive signals through engaging autonomic reflexes (autonomic control or active inference); or (iii) performing behaviour so as to alter external conditions that impact on internal homeostasis (allostasis; Gu & Fitzgerald 2014; Seth et al. 2011).
Consider an example in which blood sugar levels (an essential variable) fall towards or beyond viability thresholds, reaching unexpected and undesirable values (Gu & Fitzgerald 2014; Seth et al. 2011). Under interoceptive inference, the following responses ensue. First, interoceptive prediction error signals update top-down expectations, leading to subjective experiences of hunger or thirst (for sugary things). Because these feeling states are themselves surprising (and non-viable) in the long run, they signal prediction errors at hierarchically-higher levels, where predictive models integrate multimodal interoceptive and exteroceptive signals. These models instantiate predictions of temporal sequences of matched exteroceptive and interoceptive inputs, which flow down through the hierarchy. The resulting cascade of prediction errors can then be resolved either through autonomic control, in order to metabolize bodily fat stores (active inference), or through allostatic actions involving the external environment (i.e., finding and eating sugary things).
The sequencing and balance of these events is governed by relative precisions and their expectations. Initially, interoceptive prediction errors have high precision (weighting) given a higher-level expectation of stable homeostasis. Whether the resulting high-level prediction error engages autonomic control or allostatic behaviour (or both) depends on the precision weighting of the corresponding prediction errors. If food is readily available, consummatory actions lead to food intake (as described earlier, these actions are generated by the resolution of proprioceptive prediction errors). If not, autonomic reflexes initiate the metabolization of bodily fat stores, perhaps alongside appetitive behaviours that are predicted to lead to the availability of food, conditioned on performing these behaviours.[4]
3.2 Implications of interoceptive inference
Several interesting implications arise when considering emotion as resulting from interoceptive inference (Seth 2013). First, the theory generalizes previous “two factor” theories of emotion that see emotional content as resulting from an interaction between the perception of physiological changes (James 1894) and “higher-level” cognitive appraisal of the context within which these changes occur (Schachter & Singer 1962). Instead of distinguishing “physiological” and “cognitive” levels of description, interoceptive inference sees emotional content as resulting from the multi-layered prediction of interoceptive input spanning many levels of abstraction. Thus, interoceptive inference integrates cognition and emotion within the powerful setting of PP.
The theory also connects with influential frameworks that link interoception with decision making, notably the “somatic marker hypothesis” proposed by Antonio Damasio (1994). According to the somatic marker hypothesis, intuitive decisions are shaped by interoceptive responses (somatic markers) to potential outcomes. This idea, when placed in the context of interoceptive inference, corresponds to the guidance of behavioural (allostatic) responses towards the resolution of interoceptive prediction error (Gu & Fitzgerald 2014; Seth 2014a). It follows that intuitive decisions should be affected by the degree to which an individual maintains accurate predictive models of his or her own interoceptive states; see Dunn et al. 2010, Sokol-Hessner et al. 2014 for evidence along these lines.
There are also important implications for disorders of emotion, selfhood, and decision-making. For example, anxiety may result from the chronic persistence of interoceptive prediction errors that resist top-down suppression (Paulus & Stein 2006). Dissociative disorders like alexithymia (the inability to describe one’s own emotions), and depersonalization and derealisation (the loss of sense of reality of the self and world) may also result from dysfunctional interoceptive inference, perhaps manifest in abnormally low interoceptive precision expectations (Seth 2013; Seth et al. 2011). In terms of decision-making, it may be productive to think of addiction as resulting from dysfunctional active inference, whereby strong interoceptive priors are confirmed through action, overriding higher-order or hyper-priors relating to homeostasis and organismic integrity. It has even been suggested that autism spectrum disorders may originate in aberrant encoding of the salience or precision of interoceptive prediction errors (Quattrocki & Friston 2014). The reasoning here is that aberrant salience during development could disrupt the assimilation of interoceptive and exteroceptive cues within generative models of the “self”, which would impair a child’s ability to properly assign salience to socially relevant signals.
3.3 The predictive embodied self
The maintenance of physiological homeostasis solely through direct autonomic regulation is obviously limited: behavioural (allostatic) interactions with the world are necessary if the organism is to avoid surprising physiological states in the long run. The ability to deploy adaptive behavioural responses mandates the original Helmholtzian view of perception-as-inference, which has been the primary setting for the development of PP so far. A critical but arguably overlooked middle ground, which mediates between physiological state variables and the external environment, is the body. On one hand, the body is the material vehicle through which behaviour is expressed, permitting allostatic interactions to take place. On the other, the body is itself an essential part of the organismic system, the homeostatic integrity of which must be maintained. In addition, the experience of owning and identifying with a particular body is a key component of being a conscious self (Apps & Tsakiris 2014; Blanke & Metzinger 2009; Craig 2009; Limanowski & Blankenburg 2013; Seth 2013).
It is tempting to ask whether common predictive mechanisms could underlie not only classical exteroceptive perception (like vision) and interoception (see above), but also their integration in supporting conscious and unconscious representations of the body and self (Seth 2013). The significance of this question is underlined by realising that just as the brain has no direct access to causal structures in the external environment, it also lacks direct access to its own body. That is, given that the brain is in the business of inferring the causal sources of sensory signals, a key challenge emerges when distinguishing those signals that pertain to the body from those that originate from the external environment. A clue to how this challenge is met is that the physical body, unlike the external environment, constantly generates and receives internal input via its interoceptive and proprioceptive systems (Limanowski & Blankenburg 2013; Metzinger 2003). This suggests that the experienced body (and self) depends on the brain’s best guess of the causes of those sensory signals most likely to be “me” (Apps & Tsakiris 2014), across interoceptive, proprioceptive, and exteroceptive domains (Figure 4).
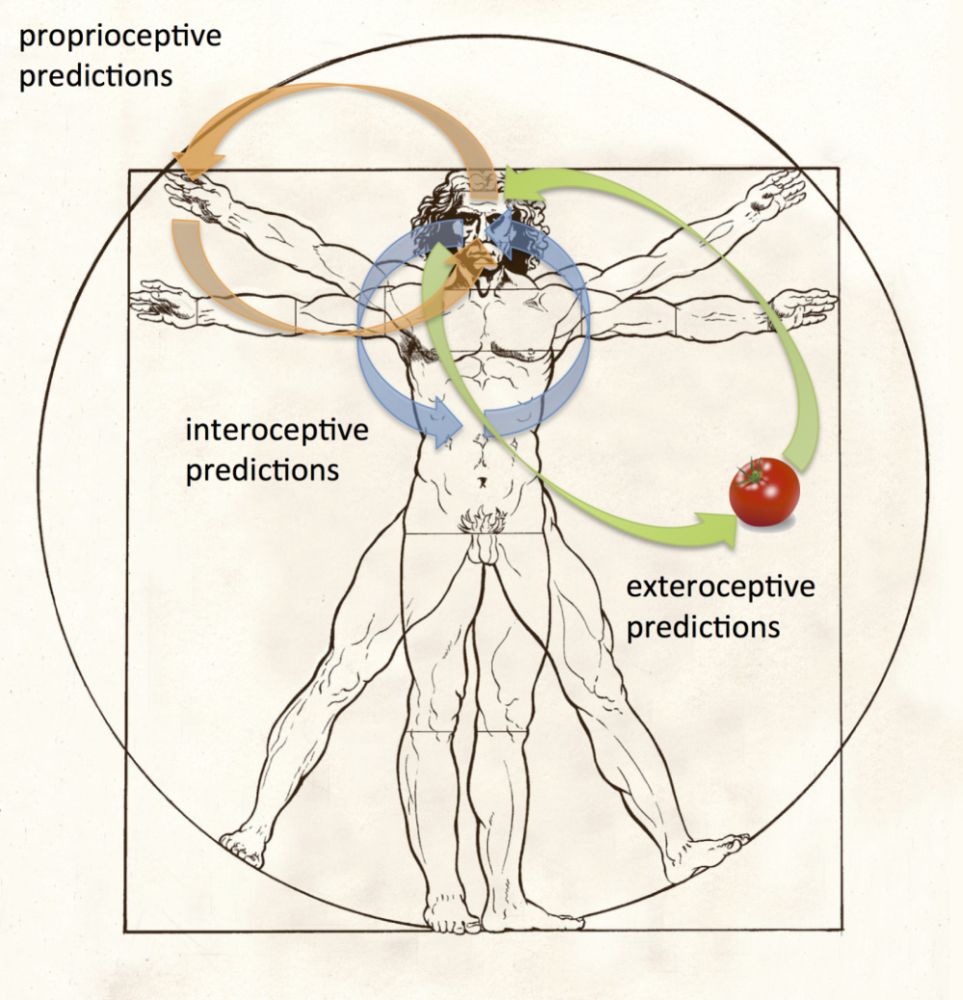 Figure 4: Inference and perception. Green arrows represent exteroceptive predictions and predictions errors underpinning perceptual content, such as the visual experience of a tomato. Orange arrows represent proprioceptive predictions (and prediction errors) underlying action and the experience of body ownership. Blue arrows represent interoceptive predictions (and prediction errors) underlying emotion, mood, and autonomic regulation. Hierarchically higher levels will deploy multimodal and even amodal predictive models spanning these domains, which are capable of generating multimodal predictions of afferent signals.
Figure 4: Inference and perception. Green arrows represent exteroceptive predictions and predictions errors underpinning perceptual content, such as the visual experience of a tomato. Orange arrows represent proprioceptive predictions (and prediction errors) underlying action and the experience of body ownership. Blue arrows represent interoceptive predictions (and prediction errors) underlying emotion, mood, and autonomic regulation. Hierarchically higher levels will deploy multimodal and even amodal predictive models spanning these domains, which are capable of generating multimodal predictions of afferent signals.
There is now considerable evidence that the experience of body ownership is highly plastic and depends on the multisensory integration of body-related signals (Apps & Tsakiris 2014; Blanke & Metzinger 2009). One classic example is the rubber hand illusion, where the stroking of an artificial hand synchronously with a participant’s real hand, while visual attention is focused on the artificial hand, leads to the experience that the artificial hand is somehow part of the body (Botvinick & Cohen 1998). According to current multisensory integration models, this change in the experience of body ownership is due to correlation between vision and touch overriding conflicting proprioceptive inputs (Makin et al. 2008). Through the lens of PP, this implies that prediction errors induced by multisensory conflicts will over time update self-related priors (Apps & Tsakiris 2014), with different signal sources (vision, touch, proprioception) each precision-weighted according to their expected reliability, and all in the setting of strong prior expectations for correlated input.[5]
While the potential for exteroceptive multisensory integration to modulate the experience of body ownership has been extensively explored both for the ownership of body parts and for the experience of ownership of the body as a whole (for reviews, see Apps & Tsakiris 2014; Blanke & Metzinger 2009), only recently has attention been paid to interactions between interoceptive and exteroceptive signals. Initial evidence in this line of investigation was indirect, for example showing correlation between susceptibility to the rubber hand illusion and individual differences in the ability to perceive interoceptive signals (“interoceptive sensitivity”, typically indexed by heartbeat detection tasks; Tsakiris et al. 2011). Other relevant studies have shown that body ownership illusions lead to temperature reductions in the corresponding body parts, perhaps reflecting altered active autonomic inference (Moseley et al. 2008; Salomon et al. 2013).
Emerging evidence now points more directly towards the predictive multisensory integration of interoceptive and exteroceptive signals in shaping the experience of body ownership. Two recent studies have taken advantage of so-called “cardio-visual synchrony” where virtual-reality representations of body parts (Suzuki et al. 2013) or the whole body (Aspell et al. 2013) are modulated by simultaneously recorded heartbeat signals, with the modulation either in-time or out-of-time with the actual heartbeat (Figure 5). These data suggest that statistical correlations between interoceptive (e.g., cardiac) and exteroceptive (e.g., visual) signals can lead to the updating of predictive models of self-related signals through (hierarchical) minimization of prediction error, just as happens for purely exteroceptive multisensory conflicts in the classic rubber hand illusion.
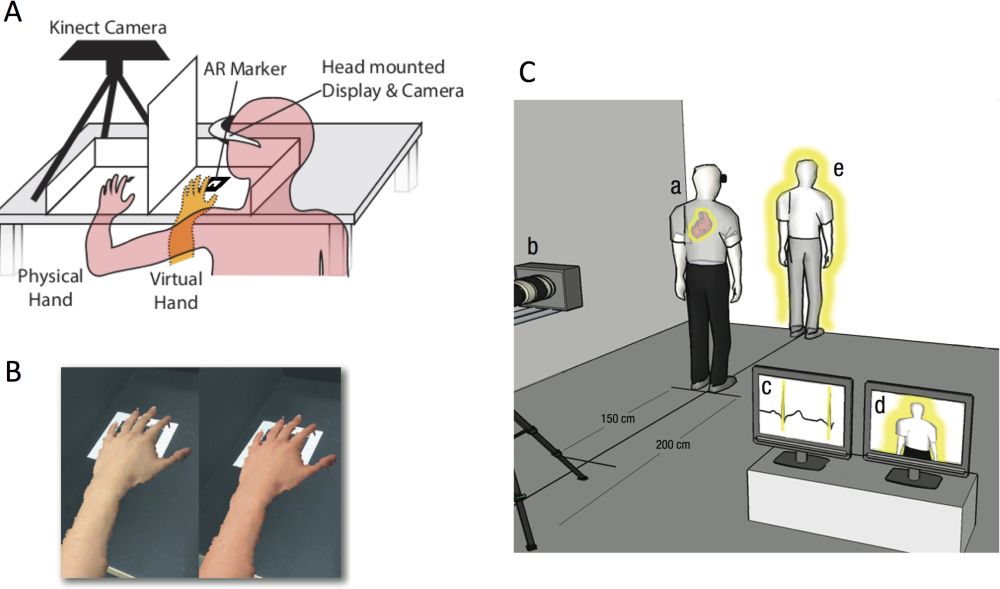 Figure 5: The interaction of interoceptive and exteroceptive signals in shaping the experience of body ownership. A. Set-up for applying cardio-visual feedback in the rubber hand illusion. A Microsoft Kinect obtains a real-time 3D model of a subject’s left hand. This is re-projected into the subject’s visual field using a head-mounted display and augmented reality (AR) software. B. The colour of the virtual hand is modulated by the subject’s heart-beat. C. A similar set-up for the full-body illusion whereby a visual image of a subject’s body is surrounded by a halo pulsing either in time or out of time with the heartbeat. Panels A and B are adapted from Suzuki et al. (2013); panel C is adapted from Aspell et al. (2013).
Figure 5: The interaction of interoceptive and exteroceptive signals in shaping the experience of body ownership. A. Set-up for applying cardio-visual feedback in the rubber hand illusion. A Microsoft Kinect obtains a real-time 3D model of a subject’s left hand. This is re-projected into the subject’s visual field using a head-mounted display and augmented reality (AR) software. B. The colour of the virtual hand is modulated by the subject’s heart-beat. C. A similar set-up for the full-body illusion whereby a visual image of a subject’s body is surrounded by a halo pulsing either in time or out of time with the heartbeat. Panels A and B are adapted from Suzuki et al. (2013); panel C is adapted from Aspell et al. (2013).
While these studies underline the plausibility of common predictive mechanisms underlying emotion, selfhood, and perception, many open questions nevertheless remain. A key challenge is to detail the underlying neural operations. Though a detailed analysis is beyond the scope of the present paper, it is worth noting that attention is increasingly focused on the insular cortex (especially its anterior parts) as a potential source of interoceptive predictions, and also as a comparator registering interoceptive prediction errors. The anterior insula has long been considered a major cortical locus for the integration of interoceptive and exteroceptive signals (Craig 2003; Singer et al. 2009); it is strongly implicated in interoceptive sensitivity (Critchley et al. 2004); it is sensitive to interoceptive prediction errors—at least in some contexts (Paulus & Stein 2006); and it has a high density of so-called “von Economo” neurons,[6] which have been frequently though circumstantially associated with consciousness and selfhood (Critchley & Seth 2012; Evrard et al. 2012).
3.4 Active inference, self-modeling, and evolutionary robotics
What role might active inference play in predictive self-modelling? Autonomic changes during illusions of body ownership (see above) are consistent with active inference; however they do not speak directly to its function. In the classic rubber hand illusion, hand or finger movements can be considered active inferential tests of self-related hypotheses. If these movements are not reflected in the “rubber hand”, the illusion is destroyed—presumably because predicted visual signals are not confirmed (Apps & Tsakiris 2014). However, if hand movements are mapped to a virtual “rubber hand”—through clever use of virtual and augmented reality—the illusion is in fact strengthened, presumably because the multisensory correlation of peri-hand visual and proprioceptive signals constitutes a more stringent test of the perceptual hypothesis of ownership of the virtual hand (Suzuki et al. 2013). This introduces the idea that active inference is not simply about confirming sensory predictions but also involves seeking “disruptive” actions that are most informative with respect to testing current predictions, and/or at disambiguating competing predictions (Gregory 1980). A nice example of how this happens in practice comes from evolutionary robotics[7]—which is obviously a very different literature, though one that inherits directly from the cybernetic tradition.
In a seminal 2006 study, Josh Bongard and colleagues described a four-legged “starfish” robot that engaged in a process much like active inference in order to model its own morphology so as to be able to control its movement and attain simple behavioural goals (Bongard et al. 2006). While there are important differences between evolutionary robotics and (active) Bayesian inference, there are also broad similarities; importantly, both can be cast in terms of model selection and optimization.
The basic cycle of events is shown in Figure 6. The robot itself is shown in the centre (A). The goal is to develop a controller capable of generating forward movement. The challenge is that the robot’s morphology is unknown to the robot itself. The system starts with a range of (generic prior) potential self-models (B), here specified by various configurations of three-dimensional physics engines. The robot performs a series of initially random actions and evaluates its candidate self-models on their ability to predict the resulting proprioceptive afferent signals. Even though all initial models will be wrong, some may be better than others. The key step comes next. The robot evaluates new candidate actions on the extent to which the current best self-models make different predictions as to their (proprioceptive) consequences. These disambiguating actions are then performed, leading to a new ranking of self-models based on their success at proprioceptive prediction. This ranking, via the evolutionary robotics methods of mutation and replication, gives rise to a new population of candidate self-models. The upshot is that the system swiftly develops accurate self-models that can be used to generate controllers enabling movement (D). An interesting feature of this process is that it is highly resilient to unexpected perturbations. For instance, if a leg is removed then proprioceptive prediction errors will immediately ensue. As a result, the system will engage in another round of self-model evolution (including the co-specification of competing self-models and disambiguating actions) until a new, accurate, self-model is regained. This revised self-model can then be used to develop a new gait, allowing movement, even given the disrupted body (E, F).[8]
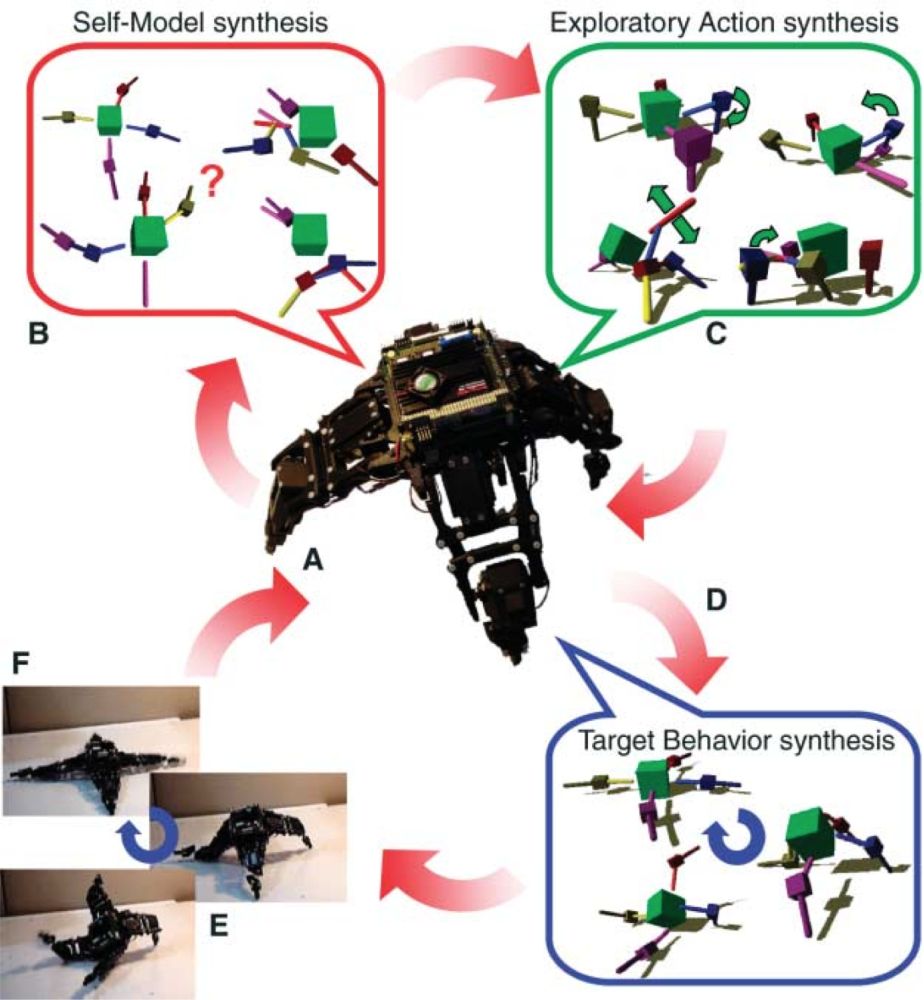 Figure 6: An evolutionary-robotics experiment demonstrating continuous self-modelling Bongardet al. (2006). See text for details. Reproduced with permission.
Figure 6: An evolutionary-robotics experiment demonstrating continuous self-modelling Bongardet al. (2006). See text for details. Reproduced with permission.
This study emphasizes that the operational criterion for a successful self-model is not so much its fidelity to the physical robot, but rather its ability to predict sensory inputs under a repertoire of actions. This underlines that predictive models are recruited for the control of behaviour (as cybernetics assumes) and not to furnish general-purpose representations of the world or the body.
The study also provides a concrete example of how actions can be performed, not to achieve some externally specified goal, but to permit inference about the system’s own physical instantiation. Bayesian or not, this implies active inference. Indeed, perhaps its most important contribution is that it highlights how active inference can prescribe disruptive or disambiguating actions that generate sensory prediction errors under competing hypotheses, and not just actions that seek to confirm sensory predictions. This recalls models of attention based on maximisation of Bayesian surprise (Itti & Baldi 2009), and is equivalent to hypothesis testing in science, where the best experiments are those concocted on the basis of being most likely to falsify a given hypothesis (disruptive) or distinguish between competing hypotheses (disambiguating). It also implies that agents encode predictions about the likely sensory consequences of a range of potential actions, allowing the selection of those actions likely to be the most disruptive or disambiguating. This concept of a counterfactually-equipped predictive model bring us nicely to our next topic: so-called enactive cognitive science and its relation to PP.